Magazine
Different types of filtration
Filtration is the purification of liquid or gaseous matter using different filtering processes. Filtration is a mechanical separation process and is exclusively physical. Terms such as “filtration” and “filter” are used synonymously in this context.
Filtration of water – focus on water hygiene
Filtration is essential for the treatment of drinking water. It is the only way to guarantee reliable protection against disease-causing microorganisms. Filtration can also remove hormones, pesticides, nitrate, nitrite, drug residues and heavy metals from the water. The choice of a particular filtration process depends on the quality of the water before treatment and the quality required of the final product (drinking water, process water, water for medical purposes).
Filters are used in the treatment of drinking water by local water providers, for example. Drinking water in Germany has to be treated in accordance with the country’s Drinking Water Ordinance (TrinkwV). More and more private households now use additional filters as well, for reasons of hygiene.
Basic principles of filtration
Filtration is divided into two basic types - surface filtration and depth filtration – according to the principle employed. A combination of both principles is known as cake filtration.
Surface filtration
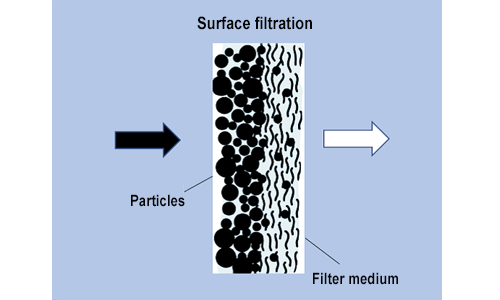
In surface filtration the particle diameter is greater than the pore diameter; this means the substances being filtered out are held back simply because of their size. The filter is usually shallow and consists of very thin membranes.
Examples: membrane filters, filter candles, microsieves, fabric filters
Membrane filtration is a technique used primarily for sterile filtration in the medical domain. With their small pore size of only 0.2 μm, membrane filters offer reliable protection against waterborne pathogens. Fabric filters are used in sewage treatment plants, for example.
Depth filtration
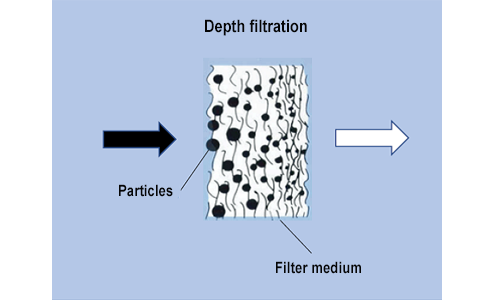
In this case the particle diameter is smaller than the pore diameter. The particles enter the pores and are held deep within the filter medium. A thick filter layer made from shaken granular materials is necessary to separate the particles from the aqueous phase. Layer thicknesses of about 1-2 metres are normally used for depth filtration.
Depth filters are typically used for applications with high levels of contamination in which other filters – such as membrane filters – do not offer satisfactory length of service life.
Cake filtration
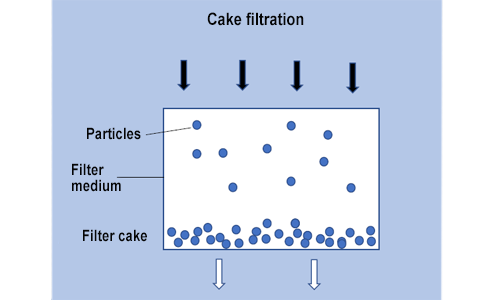
Cake filtration is a combination of surface filtration and depth filtration. Cake filtration includes all processes in which a filter cake is deposited on a filtration layer. The filter cake then either contributes to additional particle separation or itself becomes the filtering layer. Carrying out filtration requires a filter medium – one that allows the liquid to pass through while holding back solids - and a pressure difference. The filter medium can be flat (textiles and wire mesh, paper, card etc.) or it can be a layer (sand layer, sinter layer etc.). The solid material is deposited as a cake on the filter medium.
Cake filtration is used in waste water treatment plants, for example.
Special filtration processes
Membrane filtration
In the method known as membrane filtration, the water is filtered through a thin, fine-pored wall (the membrane). Depending on the nature of the membrane, this wall can hold back even the smallest particles such as bacteria and viruses. In the case of these special filters the process is called sterile filtration. Most membrane filters are made of polymer materials. In some areas, ceramic membranes and metal membranes are also used.
A distinction is made between different membrane processes such as microfiltration (MF), ultrafiltration (UF), nanofiltration (NF) and reverse osmosis (RO). The membranes in the first two processes can be seen as fine sieves that act mechanically. They are artificially manufactured membranes with a defined pore diameter. For the functioning of the membrane it is important that the pores become larger towards the pure water. In the case of nanofiltration and reverse osmosis, the membranes used are dense but allow diffusion.
The principle of reverse osmosis was first discovered in the 1950s in the desalination of sea water. This is a proven method with which to produce almost 100 % pure drinking water from sea water. In desalination, water is pressed through a synthetic semi-permeable membrane. This type of membrane is permeable only to water molecules and allows them to pass through in only in one direction.
Distinctions are made between different membrane geometries such as field membranes, hollow fibre membranes, monochannel and multichannel elements, disc and pocket membranes. Hollow fibre membranes are the type most often used to safeguard water hygiene in medical settings. They are used in the form of point-of-use filters.
Activated carbon filtration
Activated carbon filtration is essentially based on the principle of adsorption. Harmful substances such as dust and heavy metals are removed from liquids or gases with the aid of filters containing activated carbon. In the treatment of drinking water, activated carbon filters are used for the adsorptive retention of organic substances, odours and flavours. They are also used to filter drug residues out of the water.
Because this technique depends on adsorption, activated carbon has a limited capacity to take up impurities. It has to be exchanged or regenerated once its capacity has been exhausted. If activated carbon is not exchanged or regenerated in time it becomes overloaded. This can result in the filtrate containing a higher concentration of the unwanted materials than the initial impure water.
The carbon also acts as a reducing agent and is suitable for removing oxidising agents such as ozone and chlorine from spent air or wastewater. Activated carbon filters are also available with a membrane core so that the advantages of both filters can be combined.
In addition, there are various chemical and biological processes for water treatment such as disinfection and biochemical oxidation. The different processes all have their merits. The choice depends on what the water to be purified actually contains and on the quality required of the end product.